Beyond the Male Default: Sex Inclusion as a Core Principle of Biomedical Research
Implications for Scientific Validity, Patient Safety, and Policy Reform
Background and Rationale
Despite women comprising approximately 50% of the global population, biomedical research has historically been designed around the male body as the default biological model. For decades, female biology was considered a confounding variable rather than a critical determinant of health outcomes (1). This has resulted in systematic underrepresentation of women across preclinical and clinical research pipelines, creating gaps in safety, efficacy, and translational validity of medical interventions.
Biological factors such as menstruation, pregnancy, menopause, and fluctuating hormonal profiles are not peripheral considerations; they fundamentally influence pharmacokinetics, pharmacodynamics, immune responses, and disease progression. Yet, these variables are still insufficiently integrated into mainstream biomedical research design.
At the same time, it is important to acknowledge that some contemporary studies report no statistically significant sex differences for certain drugs or disease outcomes. However, such null findings often emerge from study designs that are underpowered to detect sex-specific effects, lack sex-disaggregated analysis, or are based on endpoints not sensitive to hormonal or life-stage variation (2). As a result, the absence of observed differences should not be conflated with evidence of equivalence, particularly when historical exclusion and methodological limitations persist. This raises a critical question: if a study is not designed to detect sex-specific effects, can its conclusions genuinely be considered neutral or universally applicable?
Male-Dominant Animal Models
Multiple large-scale reviews of biomedical and neuroscience literature have demonstrated a strong male bias in animal research, particularly in rodent models.
Key findings:
Approximately 60–80% of preclinical studies conducted before the early 1990s relied exclusively on male animals (3).
Even in studies where female animals were included, sex-disaggregated analyses were often not performed (2).
Neuroscience, cardiovascular, and pharmacology research showed some of the strongest male-only biases.
This foundational bias is critical, as preclinical findings directly inform dose selection, safety margins, and trial design in humans. When the underlying animal models themselves fail to adequately represent female biology, the translational validity of subsequent human trials—particularly for women—is inherently compromised (4,5). In other words, if the biological models used to generate early evidence are incomplete or biased, it becomes scientifically inconsistent to expect downstream safety and efficacy data to be robust for human females across different life stages. If the model itself is flawed, why do we continue to trust the outcome—especially when that outcome is applied to half the population?
The consequences of these limitations do not remain confined to the initial model design but extend across subsequent stages of research.

Clinical Trial Representation Across Development Stages
Although later-stage clinical trials tend to show more balanced enrollment, early-phase trials—where safety, dosing, and pharmacokinetics are established—continue to underrepresent women (6).
Estimated Representation by Research Stage (Early 1990s):
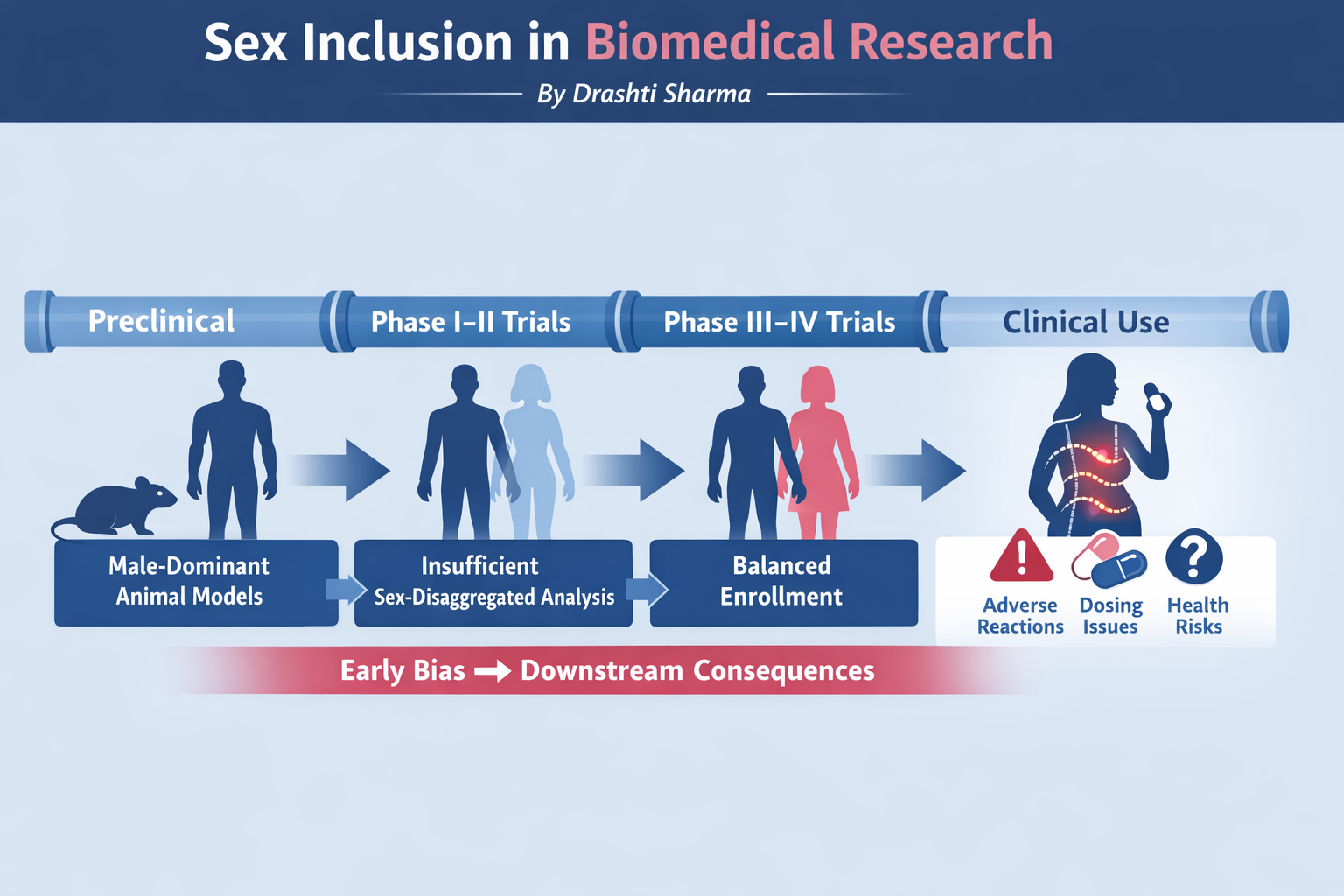
Figures represent approximate patterns reported across multiple systematic reviews and regulatory analyses rather than exact pooled estimates.
This pattern suggests that while women may be included later for efficacy endpoints, the foundational safety and dosing evidence is often derived from male-centric data. How reliable can downstream conclusions be when early safety decisions are based on incomplete biological representation
Given the persistent underrepresentation of women in early-stage trials, regulatory frameworks play a critical role in determining whether such gaps are corrected—or allowed to persist—across the research pipeline.
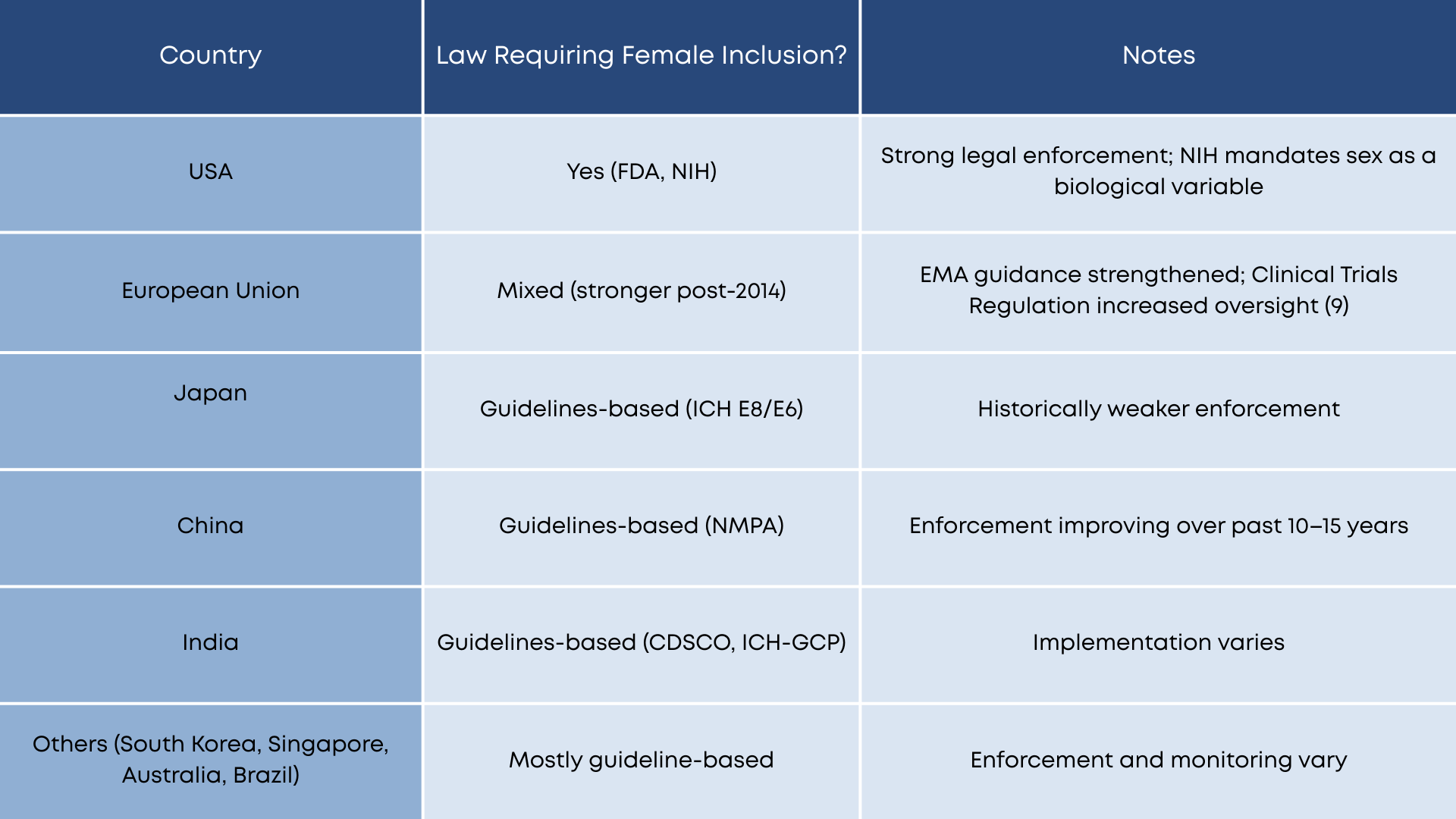
Although post-2000 reforms improved awareness, guideline-based systems often lack accountability mechanisms, resulting in inconsistent implementation.
Regulatory Landscape: Law vs Guidelines
Regulatory responses to sex-based inequities in research vary globally. While some jurisdictions have implemented legal requirements, others rely primarily on non-binding guidelines (8).
Comparative Overview:
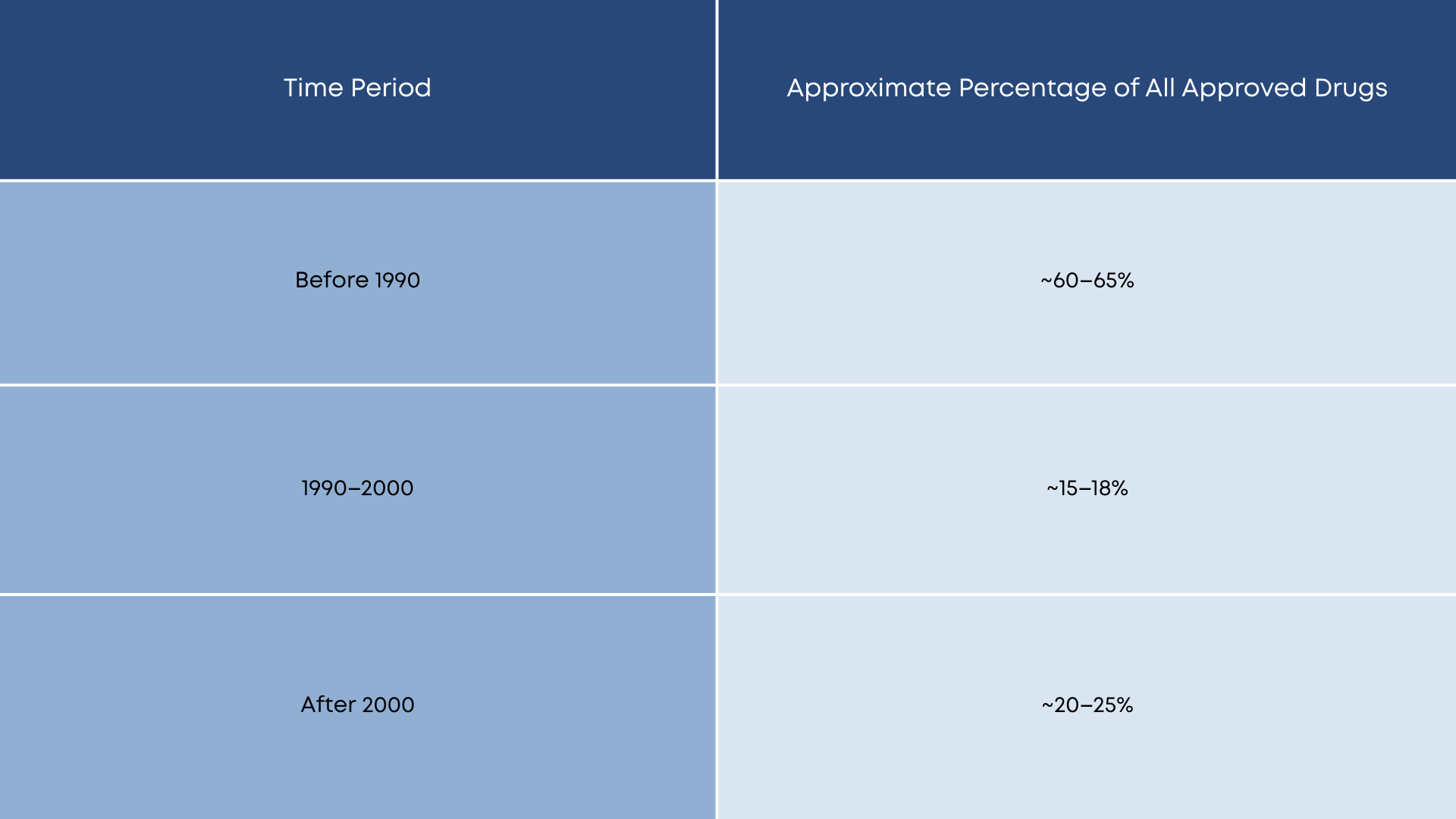
This historical context suggests that a large proportion of existing therapeutics entered the market without robust female-specific preclinical or early-phase data.
Where regulatory guidance is weak or inconsistently enforced, the consequences are not merely procedural—they manifest directly in patient-level outcomes, including drug exposure, safety, and adverse reactions.
Drug Development by Era
The majority of drugs currently in use were developed under regulatory frameworks that did not adequately mandate female inclusion (6).
Pharmacokinetics, Safety, and Adverse Outcomes
Recent analyses of FDA-approved drugs demonstrate that women frequently experience:
Higher plasma drug concentrations (1,9)
Longer elimination half-lives (1,9)
Increased incidence of adverse drug reactions (1)
In several studies, sex-specific pharmacokinetic differences predicted a higher likelihood of side effects in women, raising concerns about dose optimization and safety margins derived from male-centric data (1). If adverse outcomes are foreseeable through sex-specific analysis, why are such analyses still treated as optional rather than essential?
These risks are further amplified in populations that are systematically excluded from evidence generation altogether, most notably pregnant and breastfeeding individuals.
The Pregnancy and Lactation Evidence Gap
Pregnant and breastfeeding individuals remain one of the most excluded populations in biomedical research. Pre-approval testing in these groups is extremely rare, leading to:
Off-label medication use without evidence-based guidance
Reliance on post-marketing surveillance and observational data
Ethical and clinical uncertainty for both clinicians and patients. When evidence is missing by design, who ultimately bears the risk of that uncertainty?
Why a Comprehensive “Unsafe Drug List” Does Not Exist
A definitive list of drugs inadequately tested in women is difficult to compile due to:
Lack of sex-disaggregated reporting even when women were included
Publication bias toward null or aggregated findings
Fragmented post-marketing surveillance data
Confidentiality of regulatory dossiers
As a result, gaps often remain invisible unless adverse outcomes become clinically apparent (7).
Why Does This Issue Matters Now?
Over the past decade, biomedical research has entered a period of rapid innovation, including precision medicine, biologics, and AI-driven drug discovery. These advances promise more individualized care, yet they continue to rely on evidence pipelines historically built on male-dominant models. Without correcting foundational sex-based biases, new technologies risk reproducing old inequities at greater speed and scale.
At the same time, rising awareness of hormone-related conditions—such as autoimmune diseases, adverse drug reactions, mood disorders linked to hormonal cycles, and medication safety during pregnancy—has exposed the limitations of “one-size-fits-all” evidence. As regulatory agencies increasingly emphasize real-world evidence and post-marketing surveillance, the cost of exclusion is no longer theoretical; it manifests as preventable harm, uncertainty in clinical decision-making, and loss of public trust.
This moment therefore represents a critical inflection point: biomedical research can either integrate sex as a core scientific variable, or continue correcting harms retrospectively after drugs reach the market.
Implications for Policy and Practice
The evidence presented here suggests that improving sex inclusion in biomedical research is not merely an equity issue, but a matter of scientific validity and patient safety. Key actions include:
Strengthening enforcement of sex-based analysis requirements in preclinical and early-phase clinical research.
Requiring transparent reporting of sex-disaggregated data in regulatory submissions
Incentivizing the inclusion of pregnant and lactating populations through adaptive trial designs and post-market safeguards
Aligning funding, ethics review, and regulatory approval processes around sex as a non-optional biological variable
Addressing these gaps proactively would reduce downstream harm, improve translational accuracy, and ensure that biomedical innovation benefits all populations equitably.
Implications for Action: Moving from Evidence to Practice
The evidence presented in this brief highlights that sex-based inequities in biomedical research are not merely historical artifacts, but persistent structural issues with direct implications for patient safety, therapeutic efficacy, and public trust in medical science. Addressing these gaps requires coordinated action across research design, regulation, and knowledge translation.
Integrating Sex as a Core Scientific Variable
Sex should be treated as a fundamental biological variable across all stages of biomedical research, rather than as an optional subgroup analysis. This includes mandatory justification for single-sex studies in preclinical research and routine sex-disaggregated analysis in both animal and human studies.
Strengthening Accountability in Early-Phase Trials
Regulatory agencies and ethics committees should place greater emphasis on female inclusion in early-phase clinical trials, where safety, dosing, and pharmacokinetic parameters are established. Without representative early data, later-stage inclusion alone cannot fully mitigate risk.
Improving Transparency and Reporting Standards
Journals, funders, and regulatory bodies should require clear reporting of sex-based enrollment, analysis, and outcomes. Transparent reporting would reduce the invisibility of evidence gaps and allow clinicians and policymakers to better interpret risk.
Addressing the Pregnancy and Lactation Evidence Gap
Innovative and ethically robust research frameworks are needed to generate evidence for pregnant and breastfeeding populations, moving beyond default exclusion toward risk-informed inclusion and post-approval data integration.
Knowledge Translation and Public Engagement
Finally, translating scientific evidence into accessible formats is essential for advocacy, education, and policy reform. Clear communication of why sex-based research matters can empower patients, clinicians, and young researchers to demand higher scientific standards.
Taken together, these actions reframe sex inclusion not as a niche equity concern, but as a marker of scientific quality and translational integrity. Ensuring that biomedical research reflects biological diversity is essential for building safer, more effective, and more equitable healthcare systems.
This article was written by Drashti Sharma.
Drashti is a biomedicine undergraduate at Sunway University, Malaysia, originally from India, with a growing interest in how biological variation shapes health outcomes. Her work sits at the intersection of biomedical research, evidence gaps, and global health equity. She questions dominant research assumptions by exploring populations that fall outside “standard” biological models, and focuses on translating complex scientific issues into conversations that can inform research, policy, and advocacy.

References
Zucker, I., & Prendergast, B. J. (2020). Sex differences in pharmacokinetics predict adverse drug reactions in women. Biology of Sex Differences, 11(1), 32. https://doi.org/10.1186/s13293-020-00308-5
Flórez-Vargas, O., Brass, A., Karystianis, G., Bramhall, M., Stevens, R., Cruickshank, S., & Nenadic, G. (2016). Bias in the reporting of sex and age in biomedical research on mouse models. eLife, 5, e13615. https://doi.org/10.7554/eLife.13615
Zakiniaeiz, Y., Cosgrove, K. P., Potenza, M. N., & Mazure, C. M. (2016). Balance of the Sexes: Addressing Sex Differences in Preclinical Research. The Yale Journal of Biology and Medicine, 89(2), 255–259. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4918870/
Nunamaker, E. A., & Turner, P. V. (2023). Unmasking the Adverse Impacts of Sex Bias on Science and Research Animal Welfare. Animals : An Open Access Journal from MDPI, 13(17), 2792. https://doi.org/10.3390/ani13172792
Sex Differences, Gender Biases: What Are We Really Studying in Preclinical Research? (2025). Pharmasalmanac.com. https://www.pharmasalmanac.com/articles/sex-differences-gender-biases-what-are-we-really-studying-in-preclinical-research
Yang, Y., Carlin, A. S., Faustino, P. J., Motta, M. I., Hamad, M. L., He, R., Watanuki, Y., Pinnow, E. E., & Khan, M. A. (2009). Participation of women in clinical trials for new drugs approved by the food and drug administration in 2000-2002. Journal of Women's Health, 18(3), 303–310. https://doi.org/10.1089/jwh.2008.0971
National Academies of Sciences, Engineering, and Medicine. (1994). Women and health research: Ethical and legal issues of including women in clinical studies (Vol. 1). National Academies Press. https://nap.nationalacademies.org/read/2304/chapter/11
Ruiz Cantero, M. T., & Pardo, M. A. (2006). European Medicines Agency policies for clinical trials leave women unprotected. Journal of Epidemiology and Community Health, 60(11), 911–913. https://doi.org/10.1136/jech.2006.048769
Zucker, I., & Prendergast, B. J. (2023). Sex Differences in Pharmacokinetics. In Handbook of Experimental Pharmacology (Vol. 282, pp. 25–39). Springer. https://doi.org/10.1007/164_2023_669
